Registration and Certification Process
About the Certification Process
What is the process?
There are two linked sequential processes – Registration and Certification.
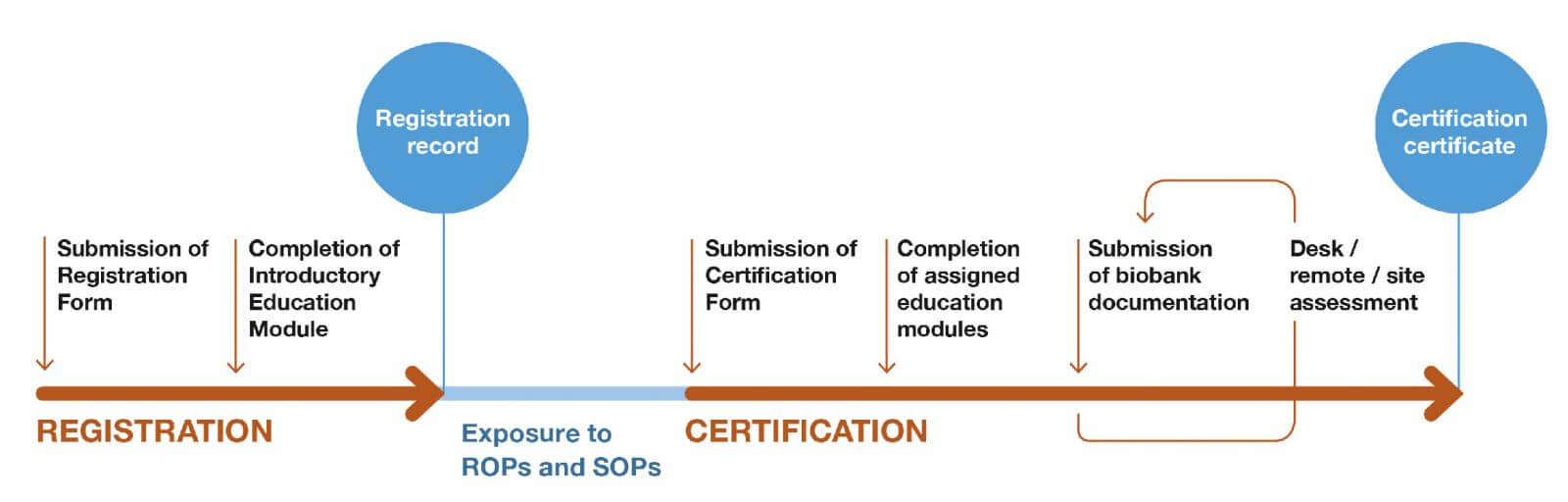
Registration: The first phase in this program. The goal is to provide the best practice-based standards and foundational knowledge for these standards.
Components:
- Biobank submits registration form and completes the on-line introductory education module ‘Basics of Biobanking’
- CTRNet provides registration record and links to the best practice-based standards.
Estimated time to completion is 2 – 3 hours.
Certification: The second phase in this program. The goals of this phase are to provide additional education modules based on the biobank type and assess biobank documents to determine if the biobank has adapted to best practice standards.
Components:
- Biobank provides additional information which enables the CTRNet Certification Office to categorize the biobank for the purpose of determining which education modules are applicable.
- Biobank submits documentation
- Verification by an objective third party (CTRNet Certification Office) that the biobank has completed education relevant to their biobank and have in place essential biobank documents.
The documents requested will be:
- REB certificate
- SOPs specific to biospecimen collection and storage
- Governance structure
- Acknowledgement declaring commitment to strive to confirm with the best practice-based standards on an ongoing basis
Templates and advice will be provided if documentation is not in place at certification application.
The effort required to complete certification will vary depending on the biobank operational complexity and number of personnel. Most biobanks will require 12 hours over one to three months of intermittent activity spread between various personnel.
How long is Certification valid?
Certification is valid for the project’s duration, to a maximum of five years. (Some biobank projects are integrated with a research project, typically with a defined duration, while other biobank projects are ongoing). However, both expire immediately if REB approval from the applicable REB(s) is not maintained or withdrawn.
Additional Activities:
- The biobank will be asked on an annual basis to update their biobank information, to identify any substantive changes that impact adherence to ROPs. If substantive changes have been made, the CTRNet Certification Office will re-review the application and re-issue the certificate.
How will my biobank be assessed?
The CTRNet Certification Office will verify that the assigned online education modules have been completed and reviewing specific required documents provided by the biobank.
The Canadian Tumour Repository Network (CTRNet) is an academic, not-for-profit consortium of Canadian tumour biobanks. The CTRNet Registration and Certification Program has been developed to address issues relating to assurance and quality in Canadian biobanking.
CTRNet uses the term “registered” to refer to biobanks that have completed the CTRNet Registration requirements. A biobank that is “registered” means that the biobank has been provided with best practice-based standards, as well as foundational knowledge for these standards, via operational practices documents and CTRNet’s web-based education course, and has completed such course.
CTRNet uses the term “certified” to refer to biobanks that, in addition to being “registered”, have completed the CTRNet Certification requirements. A biobank that is “certified” means that following “registration”, the biobank has completed the additional processes of: 1) guided self-assessment of consistency with best practice-based standards, and 2) external/peer verification that the biobank has taken measures to adapt to and/or comply with the standards on an ongoing basis.
The registration/certification of a biobank is dependent on the models and best practices employed by CTRNet, with emphasis on best practices and education.
CTRNet MAKES NO REPRESENTATION OR WARRANTY OF ANY NATURE OR KIND WHATSOEVER, EXPRESS OR IMPLIED, STATUTORY OR OTHERWISE, IN CONNECTION WITH ANY REGISTRATION/CERTIFICATION OF A BIOBANK. In providing the CTRNet Registration and Certification Program, CTRNet does not and shall not assume any responsibility, or accept or incur any obligations or liability of any nature or kind whatsoever, for any biobank which becomes “registered” or “certified” pursuant to that Program.
If you require additional information, please email certification@ctrnet.ca
Current Funders:



